(this is old code so fairly naive, and not well cleaned, but additionally you can find there GUI for windows, some tips for implementation etc.). At the beginning it was written in C++, and in given link some information with sourcesĬode is available for educational purposes If you are interested in the implementation site of IPC, the old version can be accessed here. At pH pKa p H p K a, 0.5 0.5, meaning that 50 50 of the ionizable groups in question are deprotonated. More advanced algorithm, implemented in ProMoST, takes into account localization of the charged amino acid: aaĪdditionally different pK values are used for N and C terminus depending on uncharged amino acid if applicable: As stated above, is the degree of dissociation, meaning the degree at which HX+ H X + is dissociated from the ionizable group. Each source gives different pKs.ġ Arg was not included in the study and the average pK from all other scales was takenĢ NH2 and COOH were not included in the study and they were taken from Silleroģ Bjellqvist model include also different pK values for terminal residues Unfortunately, there is no agreement in this matter. Isoelectric point determination is usage of appropriate pK values. Nevertheless, one can approximately calculate protein isoelectric point which is ± 0.5 of exact isoelectric point. Therefore, they will become cystines, which do not express any charge. Which can oxidise and form disulfide bond in protein. Problematic is also the occurrence of cysteines (negative charge) The pKa values of the side chains of the twenty common amino acids that comprise most proteins vary from approximately pH 2.8 to pH 11.2 (). 
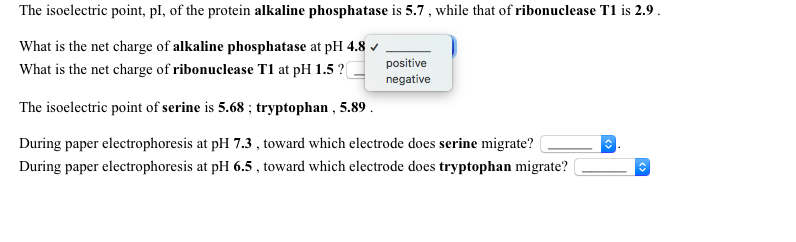
It is mainly because many proteins areĬhemically modified (amino acids can be phosphorylated, methylated, acetyleted etc.), which change their charge. The result will be almost surely different than real isoelectric point. Design of a software for calculating isoelectric point of a polypeptide according to their net charge using the graphical programming language LabVIEW. Using above formulas, we can calculate theoretical isoelectric point. In situation when the gel pH and the protein isoelectric point are equal, proteins do not move at all. 
Higher than protein isoelectric point, the particles will migrate to the anode (negative electrode) and if the buffer pH is lower than isoelectric point they will go to the cathode. For example, during electrophoresis, direction of proteins migration, depends only from their charge. Generally, macromolecules are positively charged and on the other hand, above proteins isoelectric point, 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
